Medical device companies often face challenges during biological evaluation and regulatory review, including incomplete test coverage, insufficient data depth, and gaps in immunotoxicity or blood compatibility assessments.
MDL has recently enhanced its biological evaluation service platform to address these challenges, providing more comprehensive and regulatory-aligned testing solutions for global clients.
Addressing Common Gaps in Biological Evaluation
In practice, many projects encounter issues such as:
- Incomplete mucosal irritation testing coverage
- Limited blood compatibility evaluation (e.g., hemolysis only)
- Insufficient genotoxicity data packages
- Lack of robust immunotoxicity assessment
MDL’s upgraded capabilities are designed to systematically resolve these gaps and support more robust regulatory submissions.
Comprehensive Testing Capabilities
Full-Scope Mucosal Irritation Testing
Coverage across five key mucosal types:
- Ocular
- Vaginal
- Penile
- Rectal
- Oral
![[2025 07 18] 医疗器械生物学评价服务首选mdl 6 [2025 07 18] 医疗器械生物学评价服务首选mdl 6](https://toprionbio.com/wp-content/uploads/2026/03/2025-07-18_医疗器械生物学评价服务首选MDL_6.jpg)
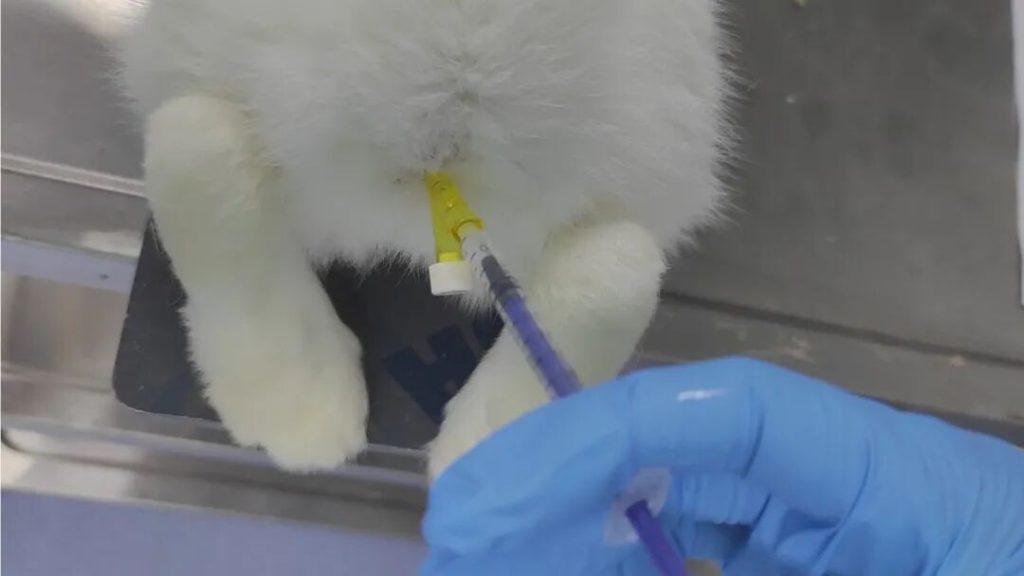
Advanced Blood Compatibility Evaluation
Beyond standard hemolysis testing, MDL provides integrated assessments including:
- Platelet activation
- Complement activation
- Coagulation pathways
- Thrombosis-related endpoints
With biomarker-level analysis such as:
- β-TG
- PF4
- TxB2
![[2025 07 18] 医疗器械生物学评价服务首选mdl 10 [2025 07 18] 医疗器械生物学评价服务首选mdl 10](https://toprionbio.com/wp-content/uploads/2026/03/2025-07-18_医疗器械生物学评价服务首选MDL_10-577x1024.jpg)
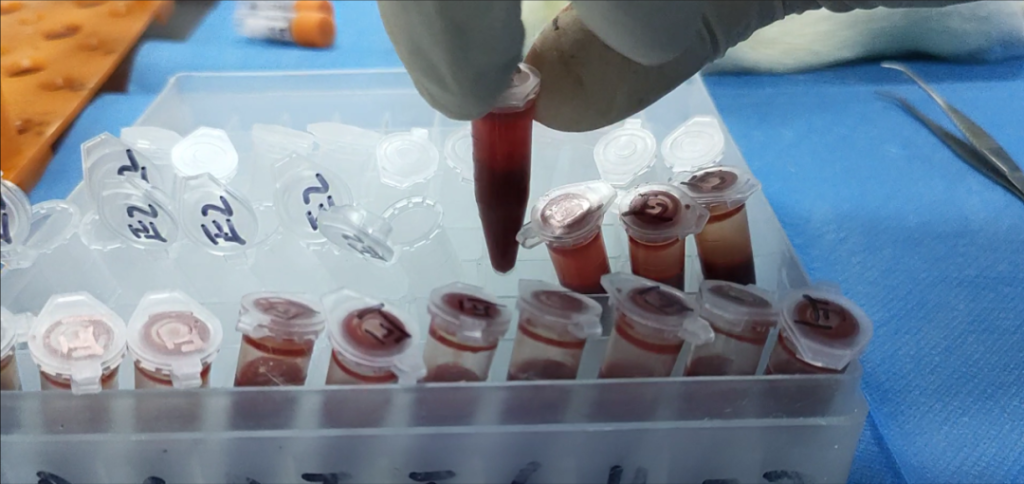
Full Genotoxicity Testing Panel
Aligned with GB/T 16886.3-2019, including:
- Ames test
- Micronucleus test
- Chromosomal aberration test
- Mouse lymphoma TK assay
![[2025 07 18] 医疗器械生物学评价服务首选mdl 12 [2025 07 18] 医疗器械生物学评价服务首选mdl 12](https://toprionbio.com/wp-content/uploads/2026/03/2025-07-18_医疗器械生物学评价服务首选MDL_12.jpg)
![[2025 07 18] 医疗器械生物学评价服务首选mdl 13 [2025 07 18] 医疗器械生物学评价服务首选mdl 13](https://toprionbio.com/wp-content/uploads/2026/03/2025-07-18_医疗器械生物学评价服务首选MDL_13.jpg)
Expanded Immunotoxicity Assessment
Based on GB/T 16886.20-2015, including:
- T lymphocyte transformation test
- Flow cytometry for lymphocyte subsets
- α-Gal antigen clearance (for animal-derived devices)
- Plaque-forming cell assay
![[2025 07 18] 医疗器械生物学评价服务首选mdl 15 [2025 07 18] 医疗器械生物学评价服务首选mdl 15](https://toprionbio.com/wp-content/uploads/2026/03/2025-07-18_医疗器械生物学评价服务首选MDL_15-1024x576.jpg)
From Basic Testing to Integrated Evaluation
Compared with conventional testing approaches, MDL provides:
- Multi-endpoint blood compatibility analysis instead of single hemolysis tests
- Advanced immunogenicity profiling beyond basic IgG/IgM detection
- Integrated evaluation strategies aligned with regulatory expectations
![[2025 07 18] 医疗器械生物学评价服务首选mdl 17 [2025 07 18] 医疗器械生物学评价服务首选mdl 17](https://toprionbio.com/wp-content/uploads/2026/03/2025-07-18_医疗器械生物学评价服务首选MDL_17-1024x683.jpg)
![[2025 07 18] 医疗器械生物学评价服务首选mdl 18 [2025 07 18] 医疗器械生物学评价服务首选mdl 18](https://toprionbio.com/wp-content/uploads/2026/03/2025-07-18_医疗器械生物学评价服务首选MDL_18-1024x683.jpg)
Global Recognition and Compliance
MDL holds both CMA and CNAS accreditations, with recognition under ILAC-MRA, ensuring:
- International acceptance of testing data
- Alignment with global regulatory requirements
- Enhanced credibility in submission dossiers
![[2025 07 18] 医疗器械生物学评价服务首选mdl 20 [2025 07 18] 医疗器械生物学评价服务首选mdl 20](https://toprionbio.com/wp-content/uploads/2026/03/2025-07-18_医疗器械生物学评价服务首选MDL_20-909x1024.png)
![[2025 07 18] 医疗器械生物学评价服务首选mdl 21 [2025 07 18] 医疗器械生物学评价服务首选mdl 21](https://toprionbio.com/wp-content/uploads/2026/03/2025-07-18_医疗器械生物学评价服务首选MDL_21-909x1024.png)
Toprion’s Role
As MDL’s international partner, Toprion provides overseas clients with direct access to CNASaccredited laboratory services. We ensure efficient communication, project coordination, and reliable delivery for global collaborations.
Looking Ahead
This accreditation reflects MDL’s ongoing commitment to delivering high-quality, compliant preclinical services aligned with global standards.